Researchers Develop a Dual-Payload Design for Targeted Cancer Therapy with Enhanced Safety
Small-molecule drug conjugates (SMDCs) are an emerging targeted cancer therapy platform with great potential. However, traditional single-payload designs have critical limitations in delivery efficiency, bystander activity and safety profile, which hamper their clinical translation and application. Current SMDC research primarily focuses on ligand optimization, the development of novel payload and the design of tumor-specific cleavable linkers. Optimizations prioritizing potency often raise on-target toxicity, whereas designs centered on selectivity may compromise therapeutic activity or restrict applicability in heterogeneous tumors. This leaves the synergetic improvement of both efficacy and safety as a core challenge in the field.
In a study published in Journal of Controlled Release, a collaborative research team led by ZHANG Xuan at the Shanghai Institute of Materia Medica (SIMM), Chinese Academy of Sciences, and HE Yonghan at the Kunming Institute of Zoology (KIZ), Chinese Academy of Sciences, proposed an innovative dual-payload design strategy for SMDC and validated its therapeutic potential in a hepatocellular carcinoma xenograft model.
The novel SMDC developed via this strategy achieved markedly improved tumor-targeting efficiency, effectively activated the bystander-killing effect, and exhibited favorable systemic applicability. CHEN Chuanjie, a joint PhD candidate at SIMM and China Pharmaceutical University, PAN Yongzhang, a PhD candidate at KIZ, and ZHANG Na, a joint master candidate at Shenyang Pharmaceutical University, contributed equally as co-first authors. ZHANG Xuan and HE Yonghan served as the corresponding authors of the study.
In recent years, the antibody-drug conjugate (ADC) field has begun to explore dual-payload or multispecific designs to boost therapeutic efficacy and overcome drug resistance. For SMDCs—which possess the unique advantages of lower molecular weight and greater structural flexibility—systematic research on such design strategies remains in its infancy. In particular, the scientific principles underlying the application of the dual-payload strategy as a universal platform to concomitantly modulate the biological effects and pharmacokinetic properties of SMDCs have not been fully elucidated. While the dual-payload strategy has shown potential to enhance efficacy and reverse drug resistance in ADCs, systematic exploration of this approach within the SMDC system has been lacking. To address this gap, the research team innovatively proposed the dual-payload strategy into SMDC design: by conjugating two therapeutic payloads to a single targeting ligand, the strategy is designed to increase payload delivery to tumor sites while limiting passive membrane permeability of the conjugate. This design is expected to enhance the selective killing of tumor cells and simultaneously reduce non-selective toxicity to normal cells, thereby synergistically broadening the therapeutic window of SMDCs. Based on XZ1606, a liver-targeting chimera (LIVTAC) developed in the previous studies, the team further designed and synthesized its dual-payload derivative, Bi-LIVTAC (XZ1618). In vitro studies demonstrated that XZ1618 exhibited superior protein degradation capacity and antiproliferative activity compared to its single-payload precursor in hepatocellular carcinoma cells with high asialoglycoprotein receptor (ASGPR) expression, and could effectively induce cell apoptosis and cycle arrest.
More importantly, using a co-culture model, this study confirmed for the first time that dual-payload SMDCs exert a prominent bystander killing effect, which can effectively act on adjacent tumor cells with low or no receptor expression and help overcome tumor heterogeneity. Meanwhile, due to the increased molecular weight and altered physicochemical properties, Bi-LIVTAC significantly inhibited passive cellular uptake in ASGPR-low-expressing cells, and its cytotoxicity showed a high degree of receptor dependence. No toxic effects were observed in multiple normal cell lines treated with Bi-LIVTAC. In the Huh-7 hepatocellular carcinoma xenograft model, Bi-LIVTAC monotherapy exerted a potent tumor-suppressive effect; furthermore, its combination with sorafenib, a first-line therapeutic agent for hepatocellular carcinoma, achieved complete tumor regression, without inducing hematological toxicities such as thrombocytopenia, demonstrating excellent systemic safety. This study not only validates the dual merits of the dual-payload strategy in improving both the efficacy and safety of SMDCs but also provides a proof of concept for its extension to other molecular targets (e.g., folate receptors) and different payload types (e.g., cytotoxins, imaging probes). It thus offers novel insights and establishes a universal platform for the development of next-generation targeted cancer therapeutics with a broader therapeutic window.
DOI: 10.1016/j.jconrel.2026.114695
Paper link: https://doi.org/10.1016/j.jconrel.2026.114695
Keywords: Small-molecule drug conjugate (SMDC); Dual-payload strategy; Therapeutic window
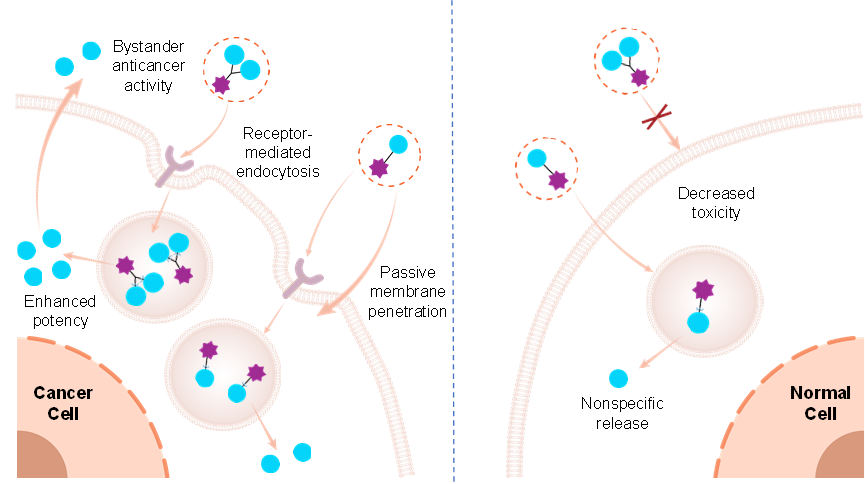
Schematic diagram of the dual-payload strategy design principle
Image by ZHANG's Lab
Contact:
DIAO Wentong
Shanghai Institute of Materia Medica
E-mail: diaowentong@simm.ac.cn




