Researchers Design an Opioid Receptor Modulator That Strengthens Morphine Analgesia at Lower Doses
Pain, especially moderate to severe chronic pain, remains a major clinical problem. Opioid drugs are still widely used because they provide strong and fast pain relief. But their benefits come with serious costs, including respiratory depression, constipation, and addiction-related risks. One strategy to address this problem is to develop positive allosteric modulators (PAMs). Unlike conventional opioids, PAMs do not compete for the main binding pocket of the receptor. Instead, they bind to a separate site and increase the receptor’s response to an opioid agonist that is already present. In principle, this approach could make it possible to achieve similar analgesia with less opioid.
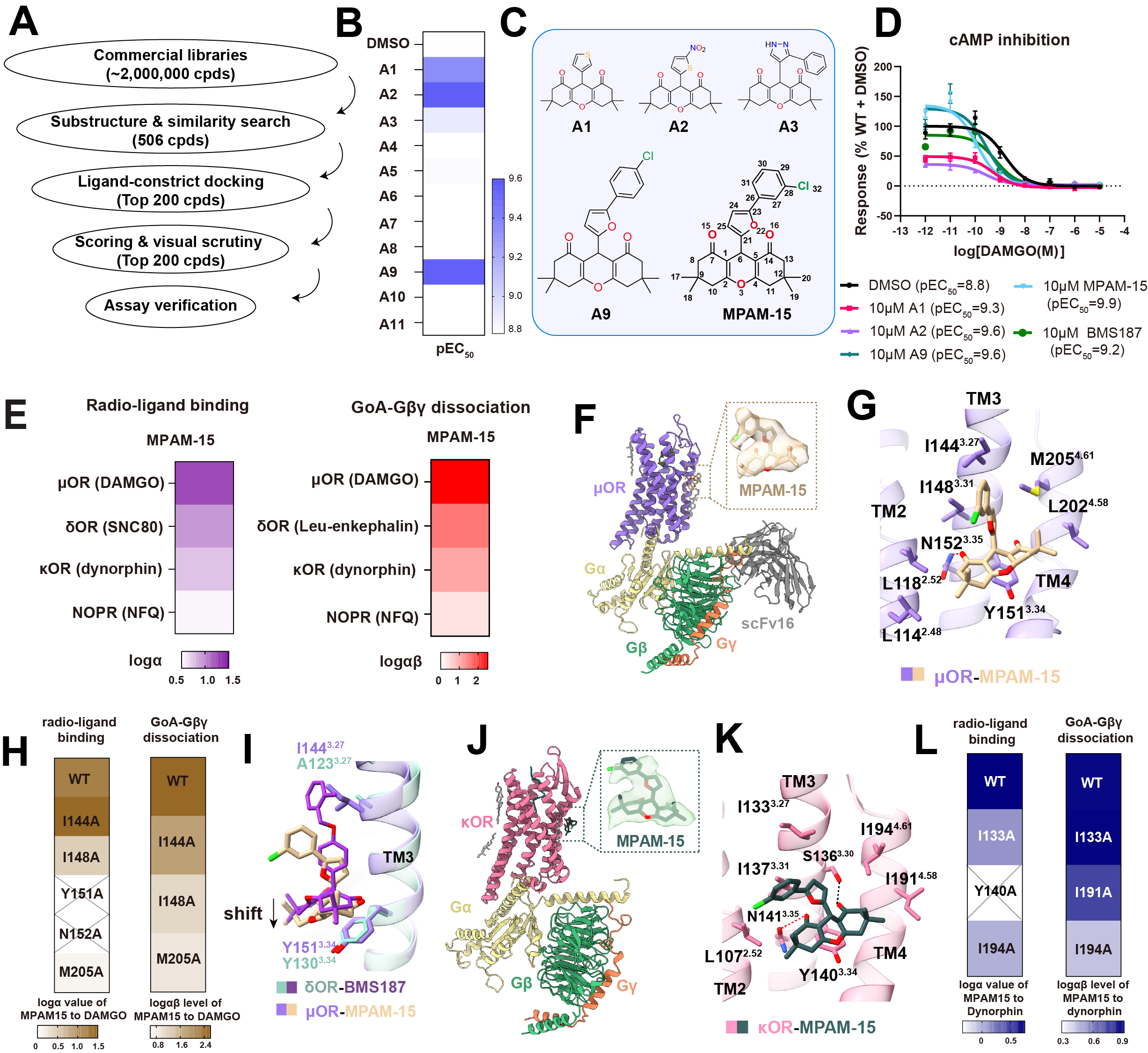
In a study published in Science Advances, a collaborative team led by XU H. Eric and GAO Zhaobing from the Shanghai Institute of Materia Medica of the Chinese Academy of Sciences, together with ZHUANG Youwen and ZHANG Jian from Shanghai Jiao Tong University and Bryan L. ROTH from the University of North Carolina at Chapel Hill, used structure-guided design to develop MPAM-15, a new modulator of the μ-opioid receptor (μOR).
The researchers found that MPAM-15 enhanced morphine-induced analgesia in mice, allowing a lower morphine dose to produce pain relief comparable to that of a higher dose. Under conditions of equivalent analgesia, the combination did not produce a matching increase in respiratory depression or constipation.
To understand how opioid receptor PAMs work, the researchers first solved high-resolution cryo-electron microscopy structures of two opioid receptor signaling complexes. These structures showed that two known PAMs, BMS-986187 and BMS-986122, did not bind in the same way, contrary to earlier pharmacology-based assumptions. The structural comparison also identified a key residue that helped explain why BMS-986187 preferred the δ-opioid receptor (δOR). Guided by these findings, the team first designed a highly δOR-selective PAM named DPAM1. They then focused on a more conserved allosteric pocket and carried out virtual screening, medicinal chemistry optimization, and functional testing to identify MPAM-15. Compared with previously reported compounds, MPAM-15 showed much stronger positive allosteric cooperativity at μOR and a clear functional preference for μOR over the other opioid receptors tested.
The researchers then solved the cryo-EM structure of the μOR-Gi-MPAM-15-DAMGO complex and found that MPAM-15 occupied the predicted allosteric site. A related structure in κ-opioid receptor (κOR) showed a similar binding mode, suggesting that this mechanism may be partly conserved across the opioid receptor family. Molecular dynamics simulations further indicated that MPAM-15 stabilized μOR in a more canonical active conformation. In simple terms, the compound appeared to shift the receptor toward a state that was better able to transmit analgesic signaling.
The team next tested whether this structural and pharmacological effect translated into a meaningful biological outcome. In several mouse pain models, MPAM-15 enhanced morphine antinociception and enabled lower-dose morphine to produce analgesia similar to that of a higher dose. Pharmacokinetic studies showed that MPAM-15 had good brain penetration, which supported its central activity. Importantly, when the researchers compared dose combinations that produced similar pain relief, they found that adding MPAM-15 to lower-dose morphine did not cause the same degree of respiratory depression or constipation seen with higher-dose morphine alone. In conditioned place preference experiments, MPAM-15 alone did not show rewarding effects, while the combination still produced morphine-related preference, although weaker than morphine alone at the same morphine dose.
The significance of this work extends beyond opioid pharmacology. By analyzing 76 class A G protein-coupled receptor (GPCR) structures that contain the conserved N3.35 residue, the researchers found that a nearby allosteric pocket may be broadly present across this receptor superfamily. This suggests that the design logic established here may not only support safer opioid analgesic development, but may also offer a transferable framework for allosteric drug discovery at other GPCR targets. Together, the study moved opioid receptor PAM research from largely empirical screening toward structure-based design and provided a practical route for developing opioid-sparing analgesic strategies.
DOI:10.1126/sciadv.aea9832
Link:https://www.science.org/doi/10.1126/sciadv.aea9832
Keywords: Opioid receptor; positive allosteric modulator; structure-guided drug design
Pharmacological characterization of MPAM-15. (Image by XU H. Eric’s lab)
Contact:
DIAO Wentong
Shanghai Institute of Materia Medica
E-mail: diaowentong@simm.ac.cn




