Scientists Unveil an Oral Drug Candidate VV261 against Chikungunya Virus
Chikungunya virus (CHIKV) is a mosquito-borne, positive-sense RNA virus that causes high fever and severe joint pain. Since 2004, CHIKV outbreaks have become more frequent and widespread. In July 2025, Guangdong province in China reported more than 9,000 confirmed cases. To date, no drug has been approved for clinical therapy.
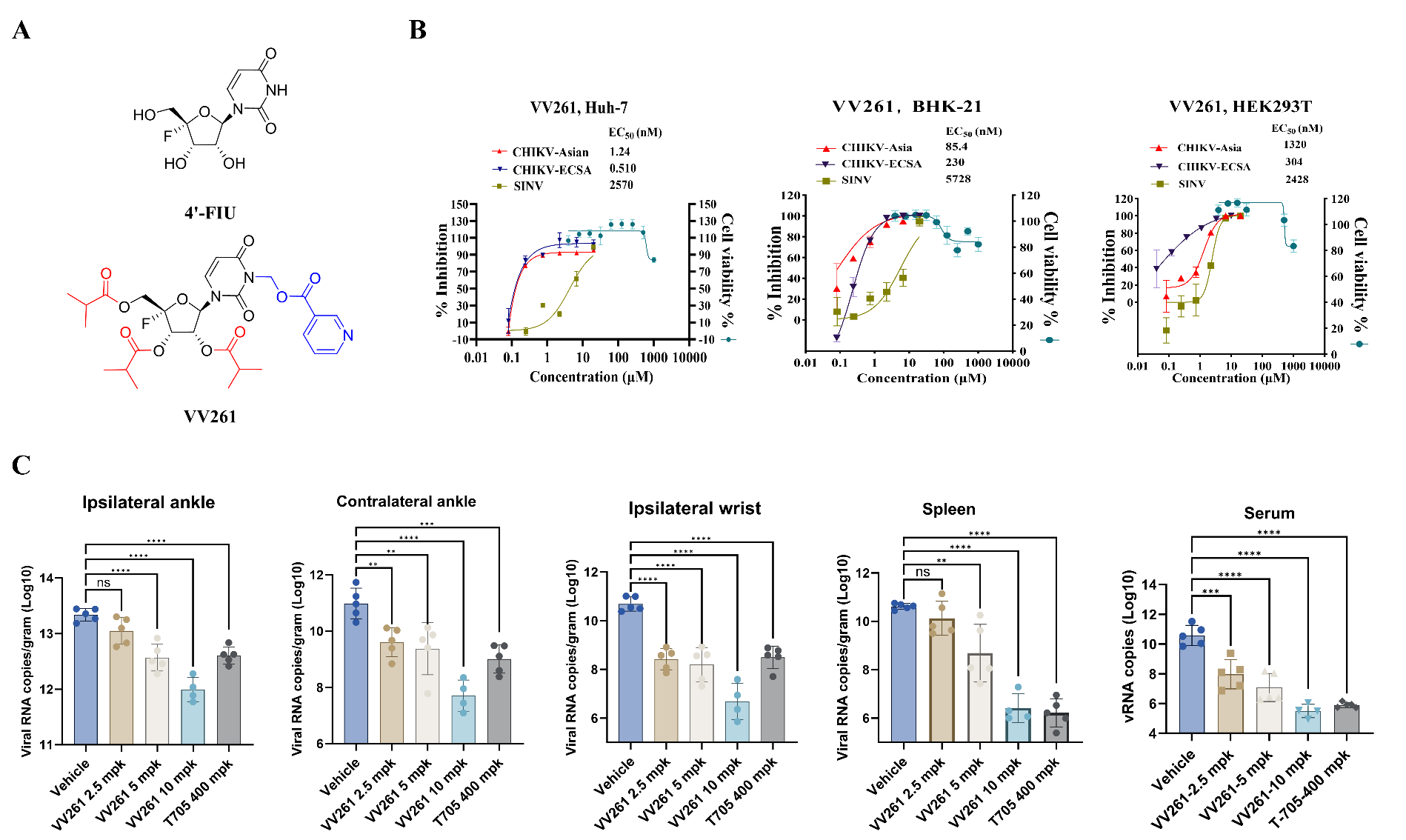
VV261 is an oral double prodrug of 4′-fluorouridine (4′-FIU), a nucleoside analog designed by SHEN Jingshan’s and XIAO Gengfu’s teams to improve stability and pharmacokinetics. It entered Phase I clinical trials in China in 2024 for severe fever with thrombocytopenia syndrome virus (SFTSV) treatment. In a study published in Signal Transduction and Targeted Therapy, the teams of XIAO Gengfu / ZHANG Leike and SHEN Jingshan reported that VV261 is a promising antiviral candidate against CHIKV.
The researchers screened a nucleos(t)ide analog library using a recombinant CHIKV-nluc infectious clone and found VV261 with remarkable antiviral efficacy. In cell culture experiments, VV261 potently inhibited CHIKV-Asian, CHIKV-ECSA, a rescued recombinant CHIKV, and Sindbis virus (SINV) across various cell lines. A Renilla luciferase (Rluc) replicon assay confirmed dose-dependent suppression of viral replication. Time-of-addition experiments indicated that VV261 acted mainly after viral entry, with the strongest effect when added 1-12 hours post infection. Uridine and cytidine partially reversed the antiviral effect, suggesting that VV261 behaved as a pyrimidine analog that interfered with nascent viral RNA synthesis. Sequencing analyses showed that VV261 did not increase lethal mutations under the tested conditions, while favipiravir (T-705) increased C-to-U transitions in progeny viral genomes.
In mice infected with CHIKV, oral administration of VV261 (2.5 mg/kg, 5 mg/kg, 10 mg/kg) significantly reduced viral RNA copies in the target tissues (such as ankle, wrist, and spleen) and serum in a dose-dependent manner. At a dose of 10 mg/kg, VV261 suppressed viral loads below the limit of detection, while T-705 required a 40-times-higher dose (400 mg/kg) to achieve a comparable reduction. Moreover, treatment with VV261 alleviated CHIKV-induced inflammation, lowering inflammatory cytokines and chemokines, including IL-6, TNF-α, IL-1β, CCL2, and CXCL10, as well as interferon-response markers such as ISG15 and IFNAR1.
This study provides solid preclinical evidence supporting VV261 as a safe and effective candidate for further clinical development against CHIKV.
Beyond CHIKV, previous studies reported that VV261 showed broad-spectrum antiviral activity against several emerging RNA viruses, including lymphocytic choriomeningitis virus (LCMV) and Crimean-Congo hemorrhagic fever virus (CCHFV), with potent inhibition in vitro and in vivo. Together with its ongoing Phase I clinical trials for SFTSV treatment, the anti-CHIKV findings reported here support further evaluation of VV261 as an oral nucleoside prodrug for other high-consequence RNA viruses.
DOI: https://doi.org/10.1038/s41392-025-02567-3
Link: https://www.nature.com/articles/s41392-025-02567-3
Keywords: Chikungunya virus, VV261, antiviral agent, nucleoside prodrug
Oral nucleoside analog VV261 showed significant anti-CHIKV efficacy both in vitro and in vivo.
(Image by SHEN Jingshan’s lab)
Contact:
DIAO Wentong
Shanghai Institute of Materia Medica
E-mail: diaowentong@simm.ac.cn




