Scientists Develop Cytoplasmic ssDNA–LNP System for Longer-Lasting Antigen Expression
Lipid nanoparticles (LNPs) have become a leading delivery platform for nucleic-acid medicines, particularly messenger RNA (mRNA). Yet mRNA can be unstable in the cytoplasm, and protein expression often declines within about 24 hours. Self-amplifying RNA (saRNA) can extend expression, but its large size makes LNP encapsulation harder. Viral-derived replicases may also heighten immune activation.
DNA is generally more chemically stable. However, conventional double-stranded DNA (dsDNA) typically depends on inefficient nuclear entry. Its rigidity and length can also reduce compatibility with LNP packaging, and it may more readily activate the cyclic GMP–AMP synthase–stimulator of interferon genes (cGAS–STING) pathway.
In a study published in the Journal of Controlled Release, researchers from the Shanghai Institute of Materia Medica, Chinese Academy of Sciences, together with teams from Lingang Laboratory, East China University of Science and Technology, and the Center for Excellence in Molecular Plant Sciences, Chinese Academy of Sciences, developed a cytoplasmic ssDNA–LNP system for longer-lasting antigen expression.
To overcome the limitations of conventional dsDNA, the research team used single-stranded DNA (single-stranded DNA, ssDNA) as a more flexible template and paired it with an orthogonal transcription enzyme, T7 RNA polymerase (T7RNAP). The goal was to enable gene expression directly in the cytoplasm, without relying on nuclear transport. This design also avoided approaches that require viral replicases or double-stranded RNA intermediates.
The platform co-delivered an ssDNA template and T7RNAP in LNPs. After cellular uptake, T7RNAP transcribed RNA from the ssDNA template in the cytoplasm. An internal ribosome entry site (IRES) supported translation, allowing expression without nuclear entry.
For proof of concept, the researchers built ssDNA vaccine constructs encoding Mpox surface antigens L1R and A33R. Mice received a single dose and were followed for 28 days. Antigen-specific immunoglobulin G (IgG) appeared by day 7. In a direct comparison, antibody levels in the mRNA group began to trend downward after day 21, while titers in the LNP–ssDNA–T7RNAP group continued to rise across the 28-day period.
Safety assessments showed activation of innate immune markers such as interferon-γ (IFN-γ) and C-X-C motif chemokine ligand 10 (CXCL10). Reported pro-inflammatory cytokines remained below thresholds associated with systemic inflammatory toxicity, and no clear liver or kidney toxicity signals were noted in the described evaluation.
DOI: 10.1016/j.jconrel.2026.114740
Link:https://www.sciencedirect.com/science/article/pii/S0168365926001422?via%3Dihub
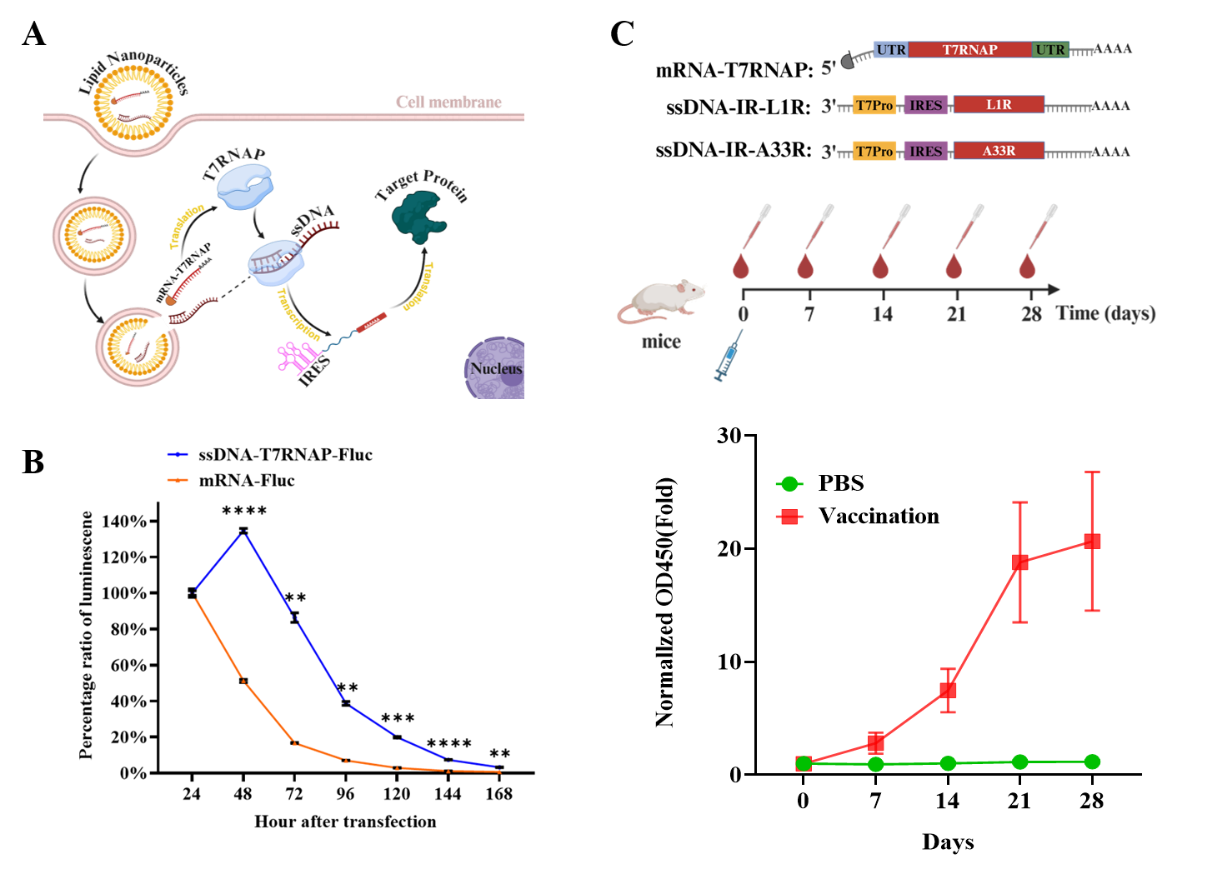
Working mechanism of the LNP–ssDNA–T7RNAP system and immunogenicity evaluation of the Mpox vaccine (Image by HAO Pei’s lab)
Contact:
DIAO Wentong
Shanghai Institute of Materia Medica
E-mail: diaowentong@simm.ac.cn




