Researchers Enable Small-Molecule-Mediated Precision Protein Editing in Living Cells
While genome editing technologies have revolutionized biology, a key gap remains: the lack of methods for direct, site-specific single amino acid editing in endogenous proteins. The ability to directly and precisely manipulate native proteins without altering their underlying genetic code represents a longstanding unmet challenge in chemical biology.
In a study published in Vita, a collaborative team of researchers led by LUO Cheng and ZHOU Hu from the Shanghai Institute of Materia Medica, together with LIN Hua from Fujian Normal University, developed LIDAPE (light-induced Asp(D)-to-Ala(A) protein editor), a modular technology designed to tackle this issue.
The approach integrates a rationally designed and screened photocatalytic warhead with a protein-binding moiety (binder), enabling site-specific conversion of aspartate to alanine at defined protein residues.
Inspired by natural enzymes, this LIDAPE platform integrates a target-specific binding module with a photocatalytic warhead. Upon visual light activation, the warhead catalyzes the hydrodecarboxylation of the proximal aspartate side chain, converting aspartate to alanine. Applying this technology to the kinase CDK2, we integrated a CDK2-specific binding moiety with the warhead. The resulting small molecule successfully achieved site-specific conversion of the critical aspartic acid (D145) to alanine (A145) with up to 86.7% efficiency, while leaving the other 15 aspartate and glutamate residues largely unchanged. Structural and computational analyses revealed that this precision is driven by a photo-induced radical hydrodecarboxylation reaction, facilitated by water molecule network rearrangement within the ATP-binding pocket. Remarkably, the editors act catalytically and function even in complex living cell environments.
The study establishes the first small-molecule platform capable of directly editing a single amino acid on an endogenous protein inside living cells. In summary, researchers have developed LIDAPEs which enable site-specific residue editing of endogenous protein in living cells, and lay the foundation for a new class of chemical biology tools. This breakthrough may elevate small-molecule drugs from traditional "physical occupation" to precise "chemical transformation", paving the way for next-generation protein-editing therapeutics.
DOI: 10.15302/vita.2026.03.0020
Link: https://www.vita-journal.com/vita/EN/10.15302/vita.2026.03.0020
Keywords: Protein Editing, Site-selective, Small Molecule
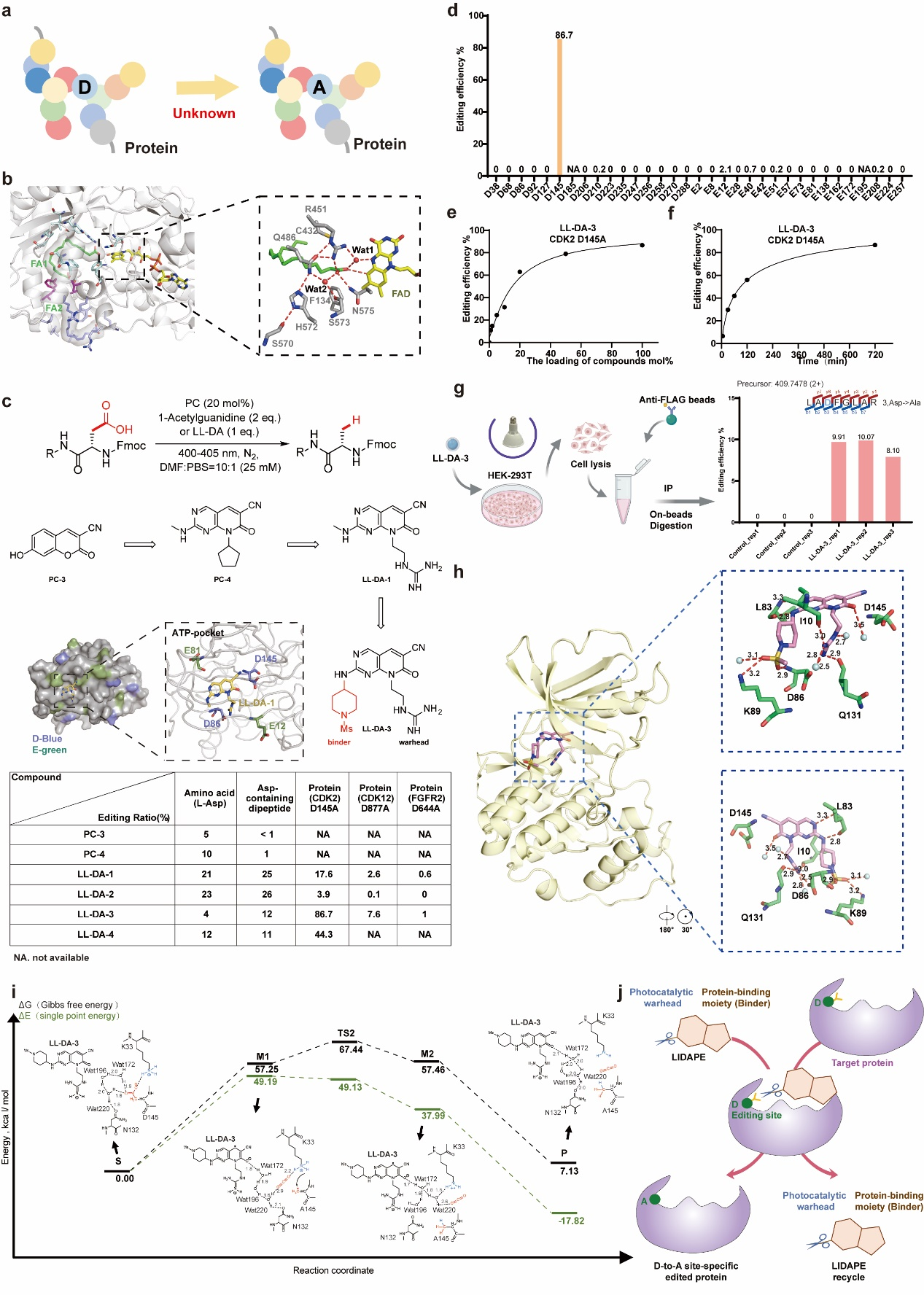
Design and characterization of the LIDAPE platform (Image by LUO Cheng's lab)
Contact:
DIAO Wentong
Shanghai Institute of Materia Medica
E-mail: diaowentong@simm.ac.cn




