Researchers Reveal Key Regulatory Elements of Lysine β-Hydroxybutyrylation
Short-chain fatty acids sit at the crossroads of multiple metabolic pathways and play important roles in diverse cellular processes. A noteworthy example is the newly identified posttranslational modification (PTM), lysine β-hydroxybutyrylation (Kbhb). Kbhb is derived from β-hydroxybutyrate, a ketone body, and histone Kbhb has shown association with active gene expression. However, key elements for regulating this physiology-relevant pathway remain unknown, hindering the functional studies of this modification in diverse biological systems. Therefore, it is necessary to characterize the mechanisms by which Kbhb exerts its biological functions.
In a study published in Science Advance on Feb. 25, Beijing time, Prof. HUANG He from Shanghai Institute of Materia Medica (SIMM) of Chinese Academy of Sciences, Prof. ZHAO Yingming from The University of Chicago and their colleagues reveal key regulatory elements of lysine β-hydroxybutyrylation.
The researchers characterized the Kbhb pathway through biochemical and proteomic approaches and identified “writer”, “eraser”, and substrate proteins for the PTM beyond a proof-of-principal demonstration of a direct function of Kbhb in transcription. "We found that the acyltransferase p300 can catalyze the enzymatic addition of β-hydroxybutyrate to lysine, while HDAC1 and HDAC2 enzymatically remove Kbhb, both in vitro and in vivo", said the researchers. They further found that p300-dependent histone Kbhb can directly mediate in vitro transcription.
In addition, the researchers carried out a proteomic screen, identifying 3248 unique Kbhb sites on 1397 proteins in HEK293 cells. These Kbhb substrates are enriched in proteins associated with diverse nuclear biological processes such as RNA metabolism, chromatin organization and DNA repair, which is quite different from the other type of PTMs such as lysine acetylation. These results indicate that different types of PTMs have unique substrate profiles, suggesting differential regulatory mechanisms.
This study therefore validates the regulation of Kbhb by pharmacologically targetable enzymes, expands the list of protein substrates and pathways potentially regulated by Kbhb, and lays the foundation necessary for further interrogation of the roles of Kbhb in various physiopathological conditions.
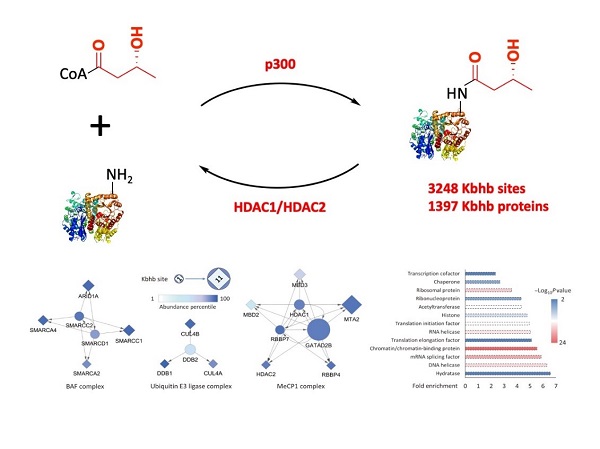
Landscape of the key regulatory elements of lysine β-hydroxybutyrylation
Link to article: https://advances.sciencemag.org/content/7/9/eabe2771
CONTACT:
DIAO Wentong
Shanghai Institute of Materia Medica, Chinese Academy of Sciences
E-mail: diaowentong@simm.ac.cn
DIAO Wentong
Shanghai Institute of Materia Medica, Chinese Academy of Sciences
E-mail: diaowentong@simm.ac.cn




