Metabotropic Glutamate Receptor Structures Uncover Mechanisms of Class C GPCR Dimerization and Activation
Metabotropic glutamate receptors (mGlus), which belong to class C G-protein-coupled receptor (GPCR) family, play key roles in modulating neuronal excitability and synaptic transmission in central nervous system. The mGlus (mGlu1-8) identified in humans serve as therapeutic targets for a variety of neurological and psychiatric disorders such as Parkinson’s disease, Alzheimer’s disease and schizophrenia, but drugs targeting the mGlus are not currently available.
In two back-to-back studies published in Nature on June 16, a team of scientists led by WU Beili, ZHAO Qiang, WANG Ming-Wei and LIU Hong at the Shanghai Institute of Materia Medica (SIMM), Chinese Academy of Sciences (CAS), in collaboration with research groups led by SUN Fei at Institute of Biophysics (IBP) of CAS and by LIU Jianfeng from Huazhong University of Science and Technology (HUST), respectively, determined six cryo-electron microscopy (cryo-EM) structures of several mGlus in distinct functional and dimeric states.
The study offers opportunities to develop new treatment for neurological and psychiatric diseases.
The six cryo-electron microscopy (cryo-EM) structures, for the first time, provide a full picture of mGlus in different conformational states, uncovering essential molecular details that modulate receptor activation and homo- and heterodimer assembly. The findings highlight the importance of the cooperativity between different subunits as well as between different receptor domains in controlling receptor function, thereby greatly expanding our knowledge about signal transduction of class C GPCRs.
The researchers solved the structures of mGlu2 and mGlu7 homodimers in inactive state, which reveal distinct dimerization modes. They also offered a thorough view of receptor conformational changes during the whole process of mGlu activation.
Importantly, this study gives valuable insights into asymmetric activation of mGlus.
Furthermore, the researchers performed structural studies on the mGlu2-mGlu7 heterodimer, and used a series of functional assays to investigate the signal transduction of the heterodimer.
Links to the papers:
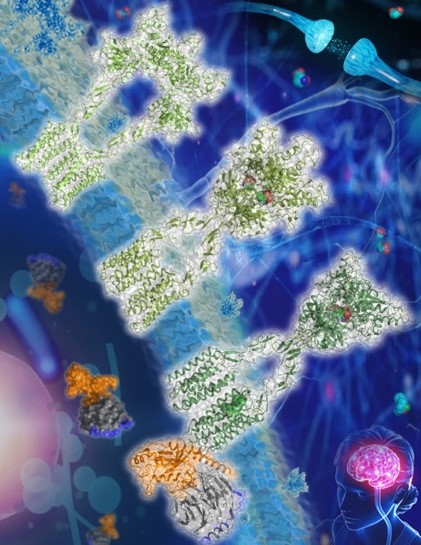
Figure: Nature reports structures of mGlu2 in different conformational states (Image by Dr. Wu Beili’s laboratory)
Contact
DIAO Wentong
Shanghai Institute of Materia Medica
E-mail: diaowentong@simm.ac.cn




