Researchers Reveal Molecular Subtypes and New Precise Therapy for KRAS Mutant Cancers
KRAS protein serves as an on/off molecular switch to relay signals from outside the cell to the cell nucleus. When the gene is mutated, KRAS will be continuously activated to promote the initiation and progression of cancers. KRAS is one of the most commonly mutated genes, which approximately presents in 20% of all human cancers. Despite its well-recognized importance in cancer, effective treatment of KRAS mutant cancer remains a major challenge for cancer therapy.
In a study recently published in Molecular Cell, a team of researchers led by TAN Minjia and HUANG Min at Shanghai Institute of Materia Medica of the Chinese Academy of Sciences, revealed new molecular subtypes of KRAS mutant cancer and identified new precise therapy.
The researchers conducted a large-scale proteomic and phosphoproteomic analysis of 43 KRAS mutant cancer cell lines across different tissue origins. By integrative analysis of the transcriptomic, proteomic, and phosphoproteomic data of these cell lines, the researchers identified three molecular subsets of KRAS mutant cancers with distinct biological characteristics. The subtyping result was further demonstrated to be useful to molecularly classify the large cohort of KRAS mutant clinical samples. Prognosis among the three subsets of clinical samples could be effectively distinguished, which demonstrated the potential clinical utility of the subtyping result.
By integrating the information from the public databases of drug sensitivity and cancer gene dependency, the researchers next investigated the subset-specific drugs, drug sensitivity biomarkers, and potential therapeutic targets. Importantly, the researchers developed a new method to predict the synergistic effects of drug combinations based on the integrative analysis of phosphoproteome and drug sensitivity information. This analysis led to the identification of a set of drug combinations with therapeutic potentials. Among these drug combinations, by using multiple cell lines and patient-derived xenograft models, they further demonstrated that the combined inhibition of the histone methyltransferase DOT1L and phosphatase SHP2 was an effective treatment for a subset of KRAS mutant cancer, corresponding to the clinical tumors with poorest prognosis.
This study provides a comprehensive proteomic and phosphoproteomic landscape of KRAS mutant cancers, inspiring a better understanding of the molecular characteristics of KRAS mutant cancer and new therapeutic possibilities.
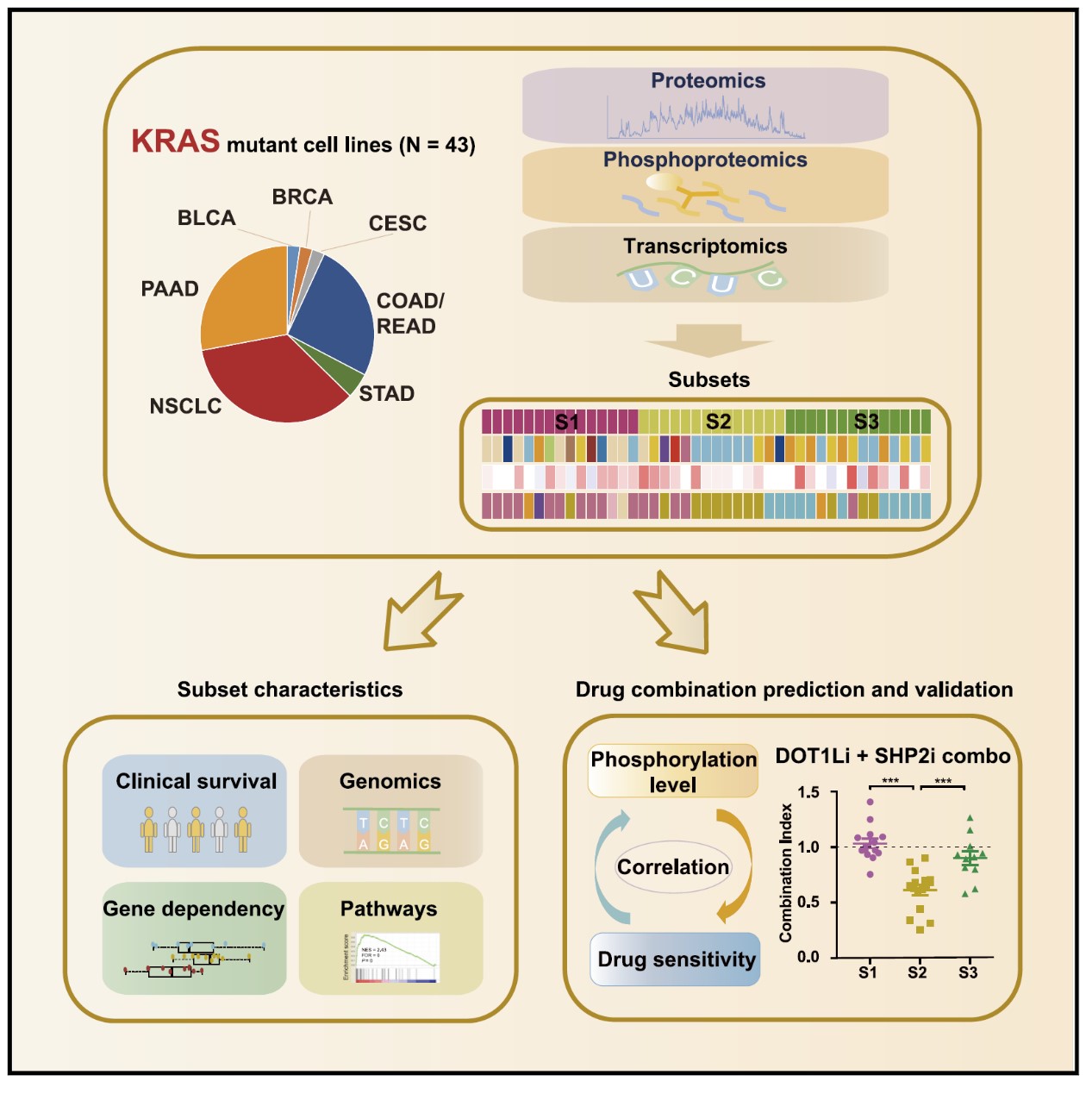
a proteomic and phosphoproteomic characterization of KRAS mutant cancers identifies combination therapies (image by Dr. Minjia Tan’s laboratory)
DOI: https://doi.org/10.1016/j.molcel.2021.07.021
Link: https://www.sciencedirect.com/science/article/abs/pii/S1097276521005906?via%3Dihub
Contact:
DIAO Wentong
Shanghai Institute of Materia Medica, Chinese Academy of Sciences
E-mail: diaowentong@simm.ac.cn




