Nuclear GTPSCS Identified as Lactyl-CoA Synthetase Promoting Histone Lactylation and Gliomagenesis
Lactate, a key product of glycolysis in mammals, is markedly elevated in pathological conditions such as tumors, sepsis, and autoimmune diseases, a phenomenon known as the Warburg effect. Histone lysine lactylation (Kla) is an epigenetic modification influenced by Warburg effect-associated L-lactate. However, the precise mechanism by which cells utilize lactate to produce lactyl-Coenzyme A (lactyl-CoA) and how this process is regulated remained unclear.
In a study published in Cell Metabolism on December 22, Prof. ZHAO Yingming from University of Chicago, Prof. HUANG He from the Shanghai Institute of Materia Medica (SIMM) of the Chinese Academy of Sciences, and their teams identified nuclear GTPSCS as a lactyl-CoA synthetase that drives histone lactylation and promotes gliomagenesis.
The researchers screened 12 fatty-acyl CoA synthetases and found that GTPSCS plays a crucial role in lactyl-CoA production and histone Kla regulation in glioma. Using mass spectrometry, they detected lactyl-CoA and determined the enzyme kinetics of GTPSCS, revealing a Michaelis constant (Km) of 15.32 ± 1.28 mM. Structural studies further showed that lactate interacts with the side chain of N308 in GTPSCS through a hydrogen bond, and mutation at N308 abolished lactyl-CoA synthetase activity.
Immunofluorescence staining revealed that nuclear localization signal (NLS) in the GTPSCS G1 subunit facilitated its nuclear translocation. Additionally, researchers found that G2 subunit residue K73 interacts with p300, forming a nuclear GTPSCS/p300 complex that regulates histone Kla, particularly at the H3K18la site, as confirmed by SILAC-based quantitative mass spectrometry.
Gliomas are the most prevalent tumors in the central nervous system. Previous studies have linked lactate dehydrogenase-dependent lactate synthesis to glioma progression, with serum lactate levels correlating with tumor grade. This study highlighted how lactate metabolism drives lactyl-CoA synthesis and histone lactylation, elucidating the molecular mechanisms involved.
Using ChIP-Seq and RNA-Seq, researchers found that H3K18la was enriched in promoters of genes like GDF15, SCX, and DYNLT4, with GDF15 strongly associated with glioblastoma prognosis and immune evasion. Analysis of The Cancer Genome Atlas (TCGA) data revealed that high expression of GDF15 and SCX correlated with poor prognosis in glioblastoma patients, while mutations in SUCLG1 and SUCLG2 were associated with reduced GDF15 expression.
Subcutaneous xenotransplantation experiments in nude mice and studies with patient-derived xenografts (PDX) demonstrated that the NLS of G1 subunit and K73ac of G2 subunit are critical for GTPSCS-mediated glioma cell proliferation and tumor progression. Immunohistochemical analysis in 170 glioma patients confirmed that H3K18la and nuclear SUCLG2 levels were higher in glioblastoma than in astrocytoma, indicating their correlation.
In summary, this study identified GTPSCS as a lactyl-CoA synthetase that catalyzes lactyl-CoA synthesis, driving histone lactylation and oncogenic gene expression in gliomas. By leveraging tumor-generated lactate, GTPSCS integrates metabolic and epigenetic pathways to promote glioma proliferation and radioresistance. These findings underscored GTPSCS as a key metabolic regulator in glioma and a potential therapeutic target for disrupting cancer-specific metabolic adaptations.
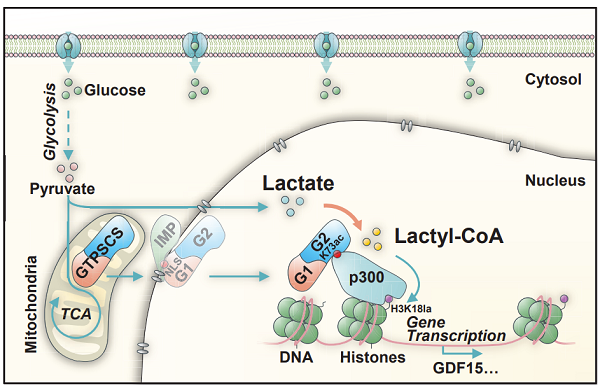
Schematic diagram of GTPSCS-mediated glioma progression through histone H3K18la (Image courtesy of ZHAO’s and HUANG’s teams).
Contact:
JIANG Qingling
Shanghai Institute of Materia Medica, Chinese Academy of Sciences
E-mail: qljiang@stimes.cn




